
A two day old male infant presents with bilious vomiting and a heart murmur. He was born to a 39-year-old G4P3 woman at 37 weeks gestation. The vital signs include T37.1 (ax), P150, R45, BP 75/50, oxygen saturation 99% in room air. Physical examination shows length, weight and head circumference at the 50th percentile. He appears jaundiced; he has a flat facial profile; epicanthal folds and upslanting palpebral fissures, flat nasal bridge, small mouth with protruding tongue, small, cupped ears, redundant nuchal skin and short hands with single palm creases. His lungs are clear to auscultation and a loud holosystolic murmur is heard. His abdomen is non-distended. Generalized hypotonia is present.
An abdominal radiograph shows a "double-bubble sign" and duodenal atresia is suspected. An echocardiogram reveals a ventricular septal defect. A chromosome abnormality is suspected and a karyotype is performed that confirms Trisomy 21.
This chapter includes some of the more common chromosome abnormalities encountered in clinical practice. The following syndromes will be reviewed: Trisomy 21 (Down syndrome), Trisomy 18 (Edwards syndrome), Trisomy 13 (Patau syndrome), XXY (Klinefelter syndrome), 45X (Turner syndrome) and Deletion 22q11.2 (Velo-Cardio-Facial syndrome/DiGeorge syndrome).
Trisomy 21 (Down syndrome)
This condition was first described clinically by Dr. John Langdon H. Down in 1866, but for almost a century the cause remained a mystery. It was not until 1959 that an extra 21st chromosome was found to be responsible for this syndrome. The CDC estimates that there is one case of Down syndrome for every 691 babies born in the United States each year.
The etiology of Down syndrome is trisomy for chromosome 21. About 95% of those with Down syndrome have standard trisomy 21 (an entire extra chromosome 21 in all cells) resulting from nondisjunction. About 90% of cases occur during maternal meiosis, predominantly in meiosis I. The other 10% occur during paternal meiosis, occurring equally between meiosis I and II. Advanced maternal age is an important risk factor for nondisjunction. Five percent of cases are due to Robertsonian translocations and mosaicism.
Robertsonian translocations in Down syndrome occur when part or all of chromosome 21 is translocated to another chromosome, usually chromosome 14. Carriers of a Robertsonian translocation are normal, but are at increased risk for miscarriage and chromosomally abnormal children. Another translocation involves chromosome 21q/21q. This is rare, but significant because a carrier parent only has one 21st chromosome (the translocated chromosome with double the genetic material). When this chromosome is present, all of the offspring will have trisomy 21.
Milder phenotypes may be seen when patients are mosaic for trisomy 21. Trisomy 21 cells are mixed with a second "normal" cell line. This can be a result of post zygotic (mitotic) nondisjunction or the post zygotic loss of a chromosome 21 from a trisomic zygote.
It is estimated that chromosome 21 contains 200 to 250 genes. The search for major genes involved in this syndrome’s characteristics is in the region 21q21–21q22.3. As technology advances, this critical region will provide further insight to our understanding of Down syndrome.
Down syndrome can usually be diagnosed at birth because of the distinctive phenotype. Clinical features include microbrachycephaly with a flat occiput, and characteristic facies including epicanthal folds, a flat nasal bridge, small mouth with protruding tongue and small ears with overfolded upper helix. The pupils may have light smudgy opaque Brushfield spots. Other features may include redundant nuchal skin, a single palm crease, and an increased space between the first and second toes. Newborns are often hypotonic and have a higher incidence of other types of malformations. Cardiac anomalies are present in 40% to 50% and include ventricular septal defects, followed by endocardial cushion defects, patent ductus arteriosus, and atrial septal defects. Gastrointestinal anomalies can include Hirschsprung disease, duodenal or anal atresia. Individuals are also at increased risk for hypothyroidism, leukemia (acute lymphocytic and nonlymphocytic leukemias), early onset Alzheimer’s disease, infectious disease susceptibility, and atlantoaxial instability which increases the risk for spinal injury.
A mnemonic for remembering the major complications found in Down syndrome is the word VALIDATE: VSD, Atlantoaxial instability, Leukemia, Immunodeficiency, Duodenal atresia, Alzheimer's disease, Thyroid dysfunction, and Endocardial cushion defects.
The mean IQ is about 45 with an upper limit of 70. The typical level of functioning is at a sixth grade level. Children can be placed in infant stimulation programs, enrolled in special education classes, and later given occupational training to help them become more independent.
When there is a high suspicion of Down syndrome based on clinical findings, a chromosome analysis (karyotype) is used to confirm the diagnosis and determine the mechanism of the trisomy. It is important to counsel parents who have one child with Down syndrome about the risk of having a second affected child. In general, the risk of recurrence is about 1%. Mothers of advanced maternal age maintain their age-related risk. A father with a balanced Robertsonian translocation has a recurrence risk of 3% to 5%, whereas a mother with a balanced Robertsonian translocation has a recurrence risk of 10% to 15%.
The American College of Obstetricians and Gynecologists (ACOG) recommends that all pregnant women be offered a screening test for Down syndrome. This includes a maternal blood test along with a special ultrasound. Women who have an abnormal screening test result are offered a diagnostic test, such as amniocentesis or chorionic villus sampling.
There is no specific treatment for Down syndrome. However, due to advances in the management of medical and surgical issues that can occur, the average lifespan has increased to almost 50 years.
Trisomy 18 (Edwards syndrome)
Infants with trisomy 18 are severely affected and usually die before birth or within the first months of life. Less than 10% of patients survive beyond twelve months and those who survive have profound developmental delay. The incidence is 1 in 4000-8000 live births, with a predominance of affected females. The etiology of this syndrome is a trisomy for chromosome 18. About 94% will have full trisomy 18. The remainder has partial trisomy 18 resulting from an unbalanced translocation or mosaicism. The clinical picture of partial and mosaic trisomy 18 vary from mild with improved survival to being indistinguishable from full trisomy 18. This depends on which part of the chromosome is affected and the degree of mosaicism.
Individuals with trisomy 18 have severe intellectual disability, intrauterine growth retardation, failure to thrive and 90% have a congenital heart malformation (such as ventricular septal defect). Physical features can include an abnormally shaped head with a prominent occiput, low-set ears, micrognathia, short sternum, characteristic clenched fists, hypoplastic toenails, misaligned toes, and "rocker-bottom feet" with a prominent calcaneus. Heart failure and central apnea contribute to infant mortality. A small number of children with trisomy 18 have reached adulthood.
The recurrence risk for full 18 trisomy is less than 1%. The risk for trisomy 18 increases with maternal age. Chromosome studies and genetic counseling are indicated. Prenatal screening is available. Women who have an abnormal screening test result are offered a diagnostic test, such as amniocentesis or chorionic villus sampling.
Treatment is supportive, but parents may elect to focus on comfort care rather than proceed with extensive treatment and resuscitative measures in a severely affected infant.
Trisomy 13 (Patau syndrome)
The constellation of findings in this condition has been described as far back as the 1600's. Klaus Patau and his colleagues were the first to attribute the syndrome of trisomy for chromosome 13 by cytogenetic analysis in 1960. The incidence is 1 in 10,000 to 20,000 live births. The etiology is usually trisomy for chromosome 13. About 5% to 10% are caused by a translocation (usually a 13;14 unbalanced Robertsonian translocation). The remainder may have a partial duplication of 13q or Trisomy 13 mosaicism. Both of these may have a milder phenotype and better outcome in regards to survival than full trisomy 13.
Newborns with Trisomy 13 are severely affected and may only survive a few days to weeks due to complex cardiac malformations or severe neurological abnormalities. Approximately 90% do not live past the first year. Individuals that do survive may have growth deficiency, severe developmental delay, and central nervous system malformations such as holoprosencephaly. Other findings may include microcephaly, microphthalmia, central apnea, seizures, hearing loss, cleft lip and palate, and genitourinary defects. Congenital cardiac anomalies, such as atrial and ventricular septal defects, are found in 80%. The hands may show single palm creases and postaxial polydactyly with overlapping of the fingers over the thumb which strongly suggest Trisomy 13.
The recurrence risk of Trisomy 13 is generally less than 1%. The risk increases with maternal age. Chromosome studies and genetic counseling are indicated. Prenatal screening is available for Trisomy 13. Women who have an abnormal screening test result are offered a diagnostic test, such as amniocentesis or chorionic villus sampling.
Treatment is supportive, but parents may elect to focus on comfort care rather than proceed with extensive treatment and resuscitative measures in a severely affected infant.
Klinefelter syndrome
Harry Fitch Klinefelter encountered a number of young male patients with small testes and gynecomastia. His description in 1942 recognized a constellation of findings which would become the syndrome that bears his name. Klinefelter syndrome affects 1 in 500 to 1000 newborn boys. The etiology is an extra X chromosome. The most common chromosomal pattern is 47,XXY, but other variants can occur including 48,XXYY, 48,XXXY, and 49,XXXXY.
In 50% to 60% of these individuals, the extra X chromosome comes from the father due to nondisjunction occurring in meiosis I. The remaining 40% to 50% comes from the mother predominantly in meiosis I. Advanced maternal age is a factor.
The characteristic findings of Klinefelter syndrome may be nonspecific in childhood. Therefore, the diagnosis is commonly made during adolescence or adulthood when pubertal maturation is delayed, incomplete, or absent. The diagnosis may also be made as part of an infertility evaluation. Chromosome analysis (karyotype) confirms the diagnosis of Klinefelter syndrome. In general, the cognitive, behavioral and physical abnormalities worsen as the number of X chromosomes increase. Those with 48,XXXY will likely be more severe than those with 47,XXY. The mildest phenotypes occur more often in those with mosaicism 46,XY/47,XXY. Characteristic features may include a tall, slender body habitus with long legs, wide hips, gynecomastia, less facial and body hair and poor muscle development. The testes are small and firm with atrophied seminiferous tubules and dysfunctional Leydig cells resulting in azoospermia and infertility. Testosterone levels are generally low while FSH and LH are elevated. Other findings may include a small penis and cryptorchidism.
Intelligence can be normal, with a mean IQ of 90. Learning and psychosocial difficulties are common. In boys who present with learning disabilities or adjustment problems at school, Klinefelter syndrome should be a consideration. Later in life, there is increased risk for diabetes mellitus, cancer of the breast, autoimmune disease, venous thromboembolism, and chronic pulmonary disease.
The management of Klinefelter syndrome includes testosterone replacement therapy which should start at the usual onset of puberty when testosterone levels are deficient and gonadotropin levels become elevated. This would allow pubertal maturation with deepening of the voice, increased muscle mass, improved bone density, penis enlargement, increased libido, improved self-esteem, and mood. With early recognition and diagnosis, treatment can be initiated to allow a more normal maturation for the affected male.
Although infertility cannot be reversed, some men with Klinefelter syndrome have minimal sperm production, therefore intracytoplasmic sperm injection (ICSI) can be an option for those wishing to have biological children. This procedure involves sperm retrieval, which is then injected directly into the egg. Offspring are at increased risk for chromosome abnormalities.
Turner syndrome
In 1938, a series of young women with short stature, webbed neck, and failure of sexual maturation were reported by Henry Turner. Eleven years later, Charles Ford determined the etiology was due to the absence of one X chromosome. Approximately 95% of 45,X fetuses die in utero. The birth incidence is 1 in 2500 live born females. Only one X chromosome is normal and functioning, the other X chromosome is not present or structurally abnormal due to deletion or translocation. About 60% to 80% of those affected retain the maternal X; thus the loss of the X chromosome is usually due to nondisjunction occurring in paternal meiosis. Advanced maternal or paternal age is not a factor in this condition.
About 50% of patients have a monosomy 45,X karyotype. Others may have a detectable mosaicism (45,X/46,XX or 45,X/46,XX/47,XXX) present. Milder phenotypes are usually seen in the mosaic karyotypes. A Y chromosome (45,X/46,XY) is present in 5% of those with Turner syndrome, and this increases the risk of gonadoblastoma.
Important characteristics to remember include short stature, webbed neck, broad chest with widely spaced nipples, amenorrhea, and coarctation of the aorta. Other findings include a small mandible, prominent ears, low posterior hair line, cubitus valgus (increased carrying angle of the elbow), short fourth metacarpal, and lymphedema of the hands and feet. The average adult height is 140 cm (4 ft., 7 in.). Cardiovascular malformations may occur in 35% to 40% and include bicuspid aortic valve and coarctation of the aorta. Other cardiac complications in adulthood include hypertension, aortic stenosis, aortic dissection, and rupture. Urinary tract malformations are found in about 30% to 40% including double collecting system, horseshoe kidney, malrotation, or abnormal vascular supply. Intelligence is usually normal; however about 10% have been found to have developmental delay.
The diagnosis is made by chromosome analysis (karyotype). Clinical suspicion may lead to testing at birth. Karyotype analysis is also warranted in the evaluation of short stature, which may be the only clinical finding present in a young girl. A karyotype is also used in the evaluation of primary amenorrhea or delayed sexual maturation.
One focus of treatment is directed at achieving normal height. Growth hormone has been successful in increasing final adult height and should be started when growth failure is detected. Anabolic steroids have an additive effect for those with extreme short stature or for older girls. The success of growth hormone therapy is determined by the age of initiation, dose, length of therapy, and inclusion of anabolic steroids.
Over 90% of those with Turner syndrome will have gonadal failure due to ovarian dysgenesis. For those who are deficient in estrogen and progestin, long term replacement therapy is required for development of secondary sexual characteristics and initiation of the menstrual cycle. Estradiol replacement therapy is recommended at 12 years of age. This allows for puberty to advance normally without interfering with final height.
Most women with Turner syndrome are infertile, although 2% to 5% may achieve spontaneous pregnancy. In vitro fertilization or other fertility treatments are available, but there is an increased risk of giving birth to a child with chromosomal or congenital anomalies. Therefore, preimplantation diagnosis is available. Genetic counseling and a medical evaluation are required.
There is a shorter life expectancy than the general population due to cardiovascular disease, hypertension, and endocrine problems. Attention to these issues can reduce morbidity and prolong life expectancy.
Deletion 22q11.2 syndrome
Other names for this condition have included Velo-Cardio-Facial syndrome, DiGeorge syndrome, Shprintzen syndrome, Conotruncal Anomaly Face syndrome, and Cayler Cardiofacial syndrome. Angelo DiGeorge described a patient in 1965 with immune deficiency and hypoparathyroidism. Shprintzen later reported patients with palatal defects, congenital heart disease, and characteristic facial features. Although previously thought to be different syndromes, a submicroscopic deletion in chromosome 22q11.2 was identified and determined as the etiology for these conditions with overlapping features. These are now designated as the Deletion 22q11.2 syndrome. Of the microdeletion syndromes, Deletion 22q11.2 is the most common. The deletion can be detected by FISH (fluorescence in situ hybridization) if the diagnosis is suspected, or by chromosomal microarray.
The incidence is 1 in 4000 live births. In about 90% of cases, there is a large 3 Mb deletion that contains about 30 genes. The remaining individuals have a smaller deletion within the DiGeorge critical region. The vast majority of cases (93%) have a de novo deletion of 22q11.2 and 7% have inherited the deletion from one parent. All those affected have a 50% chance of passing the deletion to their offspring with each pregnancy.
The classic triad includes immunodeficiency, congenital heart defects, and palatal defects. This syndrome has developmental abnormalities related to pharyngeal pouch disruption including hypoplastic parathyroid glands, hypoplastic thymus, and congenital heart defects. The clinical consequences of these anomalies can lead to hypocalcemia due to low parathyroid hormone, immunodeficiency from T-cell issues (because of the abnormal thymus), and cardiovascular malformation including tetralogy of Fallot, ventricular septal defect, other conotruncal malformations, and aortic arch abnormalities. Other findings can include cleft palate, velopharyngeal incompetence/hypernasal speech, characteristic facial features (prominent nose/nasal root, hypoplastic alae nasi, small mouth, asymmetric crying facies, ear anomalies), hearing loss, and renal abnormalities. Those affected with this syndrome can also have developmental delay including difficulties with speech. Children also have a higher risk for autism and mental health issues such as schizophrenia.
Improvements in medical care, particularly for cardiac and infectious issues, have led to longer life expectancy in patients with Deletion 22q11.2 syndrome.
Questions
1. What chromosomal disorder(s) can present with delayed or failed sexual maturation?
2. This syndrome presents with a prominent occiput, clenched fists and "rocker bottom feet". What are 2 complications that can cause death in these children?
3. Which syndrome is not associated with advanced maternal age?
. . . . . a. Down syndrome
. . . . . b. Turner syndrome
. . . . . c. Klinefelter syndrome
. . . . . d. Patau syndrome
4. True/False. The chromosomal microarray test can be used for the diagnosis in all of the genetic conditions discussed in this chapter.
5. All of the following statements regarding Turner syndrome are true EXCEPT
. . . . . a. Advanced maternal age is not a factor
. . . . . b. Most fetuses die in utero
. . . . . c. Congenital lymphedema is a common finding
. . . . . d. Most result from maternal nondisjunction
. . . . . e. All are correct
6. What causes gynecomastia in males with Klinefelter syndrome?
7. Select three features that make up the classic triad seen in patients with Deletion 22q11.2?
. . . . . a. Seizures
. . . . . b. Congenital heart disease
. . . . . c. Short stature
. . . . . d. Cleft palate
. . . . . e. Immune deficiency
Related x-rays
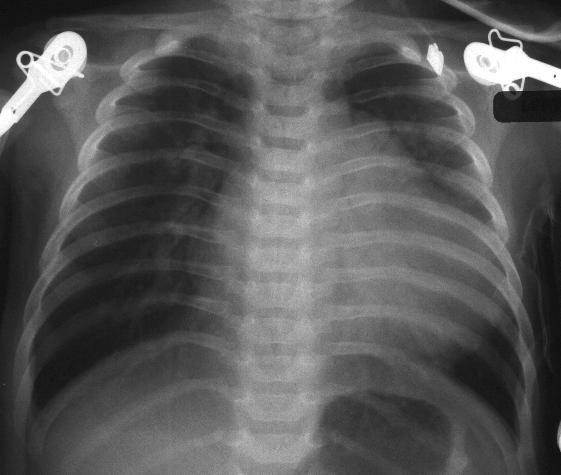
This is a portable chest X-ray from a 2 month old presenting with a "seizure" (actually tetany due to hypocalcemia), showing cardiomegaly without the expected thymic shadow (i.e., a presentation of DiGeorge syndrome) from: Yamamoto LG. Seizure and VSD in a 2-month old Infant. In: Yamamoto LG, Inaba AS, DiMauro R (eds). Radiology Cases In Pediatric Emergency Medicine, 1995, volume 2, case 2. Read the online case and description at: www.hawaii.edu/medicine/pediatrics/pemxray/v2c02.html

References
1. Bondy CA for the Turner Syndrome Consensus Study Group. Care of girls and Women with Turner Syndrome: A Guideline of the Turner Syndrome Study Group. J Clin Endocrinol Metab 2007;92(1):10-25.
2. Bull MJ and the Committee on Genetics. Health Supervision for Children with Down Syndrome. Pediatrics 2011;128:393-406.
3. Cassidy SB, Allanson JE. (Eds). Management of Genetic Syndromes. Third edition, 2010, New Jersey: John Wiley & Sons, Inc.
4. Firth H, Hurst JA. Oxford Desk Reference Clinical Genetics. 2006, New York: Oxford University Press.
5. Gorlin RJ, Cohen MM Jr. Syndromes of the Head and Neck. 2001, New York: Oxford University Press.
6. Jones KL (Ed). Smith's Recognizable Patterns of Human Malformation, Sixth edition, 2006, Philadelphia: Elsevier Saunders Company.
7. Parker SE, Mai CT, Canfield MA, et al. Updated National Birth Prevalence Estimates for Selected Birth Defects in the United States, 2004-2006. Birth Defects Res A. 2010;88:1008-1016.
8. Saller DN Jr., Canick JA. Current methods of prenatal screening for Down syndrome and other fetal abnormalities. Clin Obstet Gynecol. 2008;51(1):24-36.
Answers
1. Turner syndrome and Klinefelter syndrome
2. Trisomy 18. Heart and respiratory failure
3. b. Turner syndrome.
4. True, although all but 22q11.2 deletion syndrome can be detected by routine karyotype.
5. d
6. Elevated estradiol to testosterone ratio
7. b,d,e. Tetany due to hypocalcemia occurs, and these resemble seizures.