
The editors and current authors would like to thank and acknowledge the significant contribution of the previous author of this chapter from the 2004 first edition, Dr. Lance K. Shirai. This current second edition chapter is a revision and update of the original author’s work.
This is a 2 month-old male who presents to the emergency room with a chief complaint of fever, lethargy, and poor feeding for the past 36 hours. His parents began noticing increasing lethargy and tiring with feeding and increased work of breathing for about 12 hours prior to presentation.
He is the product of a G2P1, full term, uncomplicated pregnancy and spontaneous vaginal delivery. Nursery course was uneventful.
Exam: VS T 37.4C, HR 240, RR 72, BP 87/64, oxygen saturation 98% in room air. He is well developed, well nourished, but pale, lethargic and tachypneic, with mild subcostal retractions. HEENT exam is normal. Neck is supple without adenopathy. Lungs have good aeration with fine crackles and mild retractions. His heart is tachycardic with a regular rhythm. No murmur, rub, or valve clicks are heard. His abdomen is soft, non-distended, non-tender, and without masses. His liver is 2 to 3 cm below right costal margin. His feet and hands are cool. His peripheral pulses are 1+ to 2+ (out of 4+) throughout. Capillary refill time is 3 to 4 seconds. He has no rashes or other significant lesions.
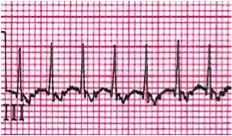
A chest x-ray shows mild cardiomegaly and mild pulmonary edema. A 12 lead electrocardiogram shows a narrow complex tachycardia (rate of 240 bpm) with no visible P-waves (rhythm strip below).

The patient is felt to be in supraventricular tachycardia and mild congestive heart failure. A peripheral IV is started and he is given a rapid IV bolus dose of adenosine. The patient immediately becomes briefly bradycardic followed by resumption of a normal sinus rhythm at a rate of 140 beats per minute. He is admitted for overnight observation and initiation of an anti-arrhythmic medication. A 12-lead electrocardiogram (ECG) following conversion shows no evidence of a delta-wave, so he is started on digoxin.
An arrhythmia (irregular heart rhythm) is not an unusual finding in children with or without known cardiac disease. Some arrhythmias are normal findings in healthy children, such as premature atrial contractions and sinus arrhythmia, however most of the arrhythmias discussed in this chapter require some sort of intervention or treatment. Most children can be satisfactorily evaluated with a history, physical exam, 12-lead ECG and rhythm strip, with possible supplementation by a chest x-ray, echocardiogram, Holter or event monitor, or an exercise study. There are several important determinants of arrhythmias, which should be considered. These include the arrhythmogenic substrate (e.g., accessory conduction pathway with reentry circuit, automatic ectopic focus), modulating factors, and triggers of the arrhythmia.
There are many ways of classifying arrhythmias. We can classify them based on the rate: tachyarrhythmias are fast, bradyarrhythmias are slow. We can also group them based on the origin of the arrhythmia: supraventricular versus ventricular. Supraventricular tachycardia (SVT), the diagnosis in this case, will be discussed first, with other pertinent arrhythmias discussed later in this section.
SVT is a tachyarrhythmia, usually manifested by a narrow complex QRS (<0.08 sec), originating above the bundle of His (i.e., including atria, sinoatrial node and atrioventricular node). There are seventeen different kinds of supraventricular tachycardias including sinus tachycardia, junctional tachycardia, atrial flutter and atrioventricular reentrant tachycardia.
Supraventricular tachycardia is the most common tachyarrhythmia in the pediatric age group, estimated to occur in approximately 1 to 4 per 1000 children. The most common types of SVT in children include atrioventricular reentrant tachycardia (AVRT), which includes Wolff-Parkinson-White syndrome (WPW), and AV nodal reentrant tachycardia (AVNRT). Overall, the incidence of AVRT is more common in children, with AVNRT increasing into young adulthood. In patients with structural heart disease (congenital or acquired), the incidence of SVT increases to 9%-37%. 50% to 60% of cases occur prior to 1 year of age, with later peaks occurring at 6-9 years old and in adolescence. While statistics vary, the spontaneous resolution rate is much higher prior to 1 year of age than if it persists past this age.
AVRT and AVNRT are considered reciprocating tachycardias, with the presence of two discrete pathways. During the tachycardia, the electrical circuit consists of a loop created by two pathways: one with antegrade conduction and the other with retrograde conduction. One pathway is considered a "fast" pathway, with rapid conduction, and the other a "slow" pathway, with slower conduction. In AVRT with orthodromic conduction, antegrade conduction occurs down the AV node pathway, which is usually the "slow" pathway, and retrograde conduction occurs up the accessory conduction tissue, which is usually the "fast" pathway. Antidromic conduction is characterized by antegrade conduction down the "fast" accessory pathway, and retrograde conduction up the "slow" AV nodal pathway (this is less common, occuring in about 5% of AVRT). In AVNRT, both pathways are found in the AV node, whereas, in AVRT, the pathway is located externally to the AV node (i.e., accessory pathway in WPW).
AVRT or AVNRT usually has its onset at rest, but may initiate during exercise. The precipitating factor(s) is often difficult to identify, but occasionally a febrile illness may precipitate an episode. The heart rate is usually in the 160 to 300 beats/min range. In general, the younger the patient the more rapid the SVT heart rate, but the longer the tachycardia is tolerated before symptoms (see below) become obvious. Typically, episodes of SVT occur and terminate abruptly, and may last anywhere from minutes to hours.
In infants, symptoms may not become apparent until the patient has been in SVT for 24 hours, or longer. They will often present with symptoms of congestive heart failure such as tachypnea, pallor, poor feeding, fussiness or lethargy, just as in our patient.
In children and adolescents, symptoms may include palpitations, chest pain, shortness of breath, dizziness, pallor, and diaphoresis. It is unusual for older patients to present in heart failure, as they are able to verbalize a "funny feeling in their chest, a "racing heart", or other symptoms soon after the onset of SVT. Syncope usually does not occur, but in any setting where cardiac output, and, therefore, cerebral perfusion pressure is decreased, it is a possibility.
As mentioned above, the differential diagnosis of a pediatric patient who presents with a narrow complex tachycardia includes AVRT, AVNRT, sinus tachycardia, atrial flutter, atrial fibrillation, junctional ectopic tachycardia, atrial ectopic tachycardia, and multifocal atrial tachycardia/chaotic atrial tachycardia.
It is important to note that some patients with SVT and a bundle branch block or antidromic WPW, may present with a wide complex tachycardia, which if often difficult to distinguish from ventricular tachycardia (VT).
Most of the narrow complex tachyarrhythmias may be distinguished by their electrocardiogram findings. AVRT ranges in heart rate from 160 to 300 beats per minute. AVNRT (Figure 1) is usually somewhat slower, in the range of 140-220 bpm. The diagnosis of AVRT or AVNRT requires the presence of 1:1 A-V conduction. The heart rate usually remains in a very narrow range regardless of the patient's physiologic state. P-waves, which are oftentimes retrograde, are visible in about 80% of cases, but may be hidden in QRS complexes. Upon conversion to a sinus rhythm, patients with WPW or Mahaim fibers (an accessory pathway able to conduct only in an antegrade fashion, connecting the AV node directly to a portion of the right bundle) will demonstrate the classical delta wave (Figure 2) as evidenced by an upsloping or slurring of the initial portion of the QRS complex. Delta waves are secondary to rapid antegrade conduction from the atrium or AV node to the ventricles through the accessory pathway, thus causing ventricular pre-excitation. With WPW, the PR interval is short, but with the presence of Mahaim fibers the PR interval is normal. Forms of SVT with concealed accessory pathways (i.e., those capable of only retrograde conduction), will not show evidence of a delta wave, and therefore most will have normal PR intervals. Most patients with AVRT and AVNRT have normal cardiac anatomy, with atrial fibrillation (AFib) and atrial flutter (AFlutter) being more common in structural heart disease. Congenital heart defects in which AVRT is most commonly encountered are Ebstein's anomaly and L-transposition of the great arteries.
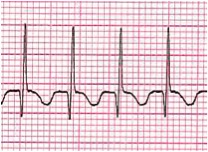
Figure 1. AVNRT
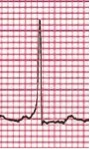
Figure 2. Delta Wave in WPW
The management approach for SVT depends upon the age and condition of the patient on presentation as recommended in the the PALS (Pediatric Advanced Life Support) algorithms. Short-term goals include stabilizing the patient, terminating the SVT and establishing a mechanism. If possible, always do an ECG with rhythm strip prior to and during termination of the tachycardia. If the patient is clinically stable, vagal maneuvers may be initially attempted to convert the tachycardia. Such vagal maneuvers may include bearing down (as though having a bowel movement (i.e., Valsalva maneuver)), blowing in a straw or inducing the diving reflex using an ice bag to the face for infants. Other vagal maneuvers such as eyeball pressure and unilateral carotid massage are harmful and should not be performed.
If the patient appears clinically unstable, urgent electrical cardioversion is indicated using 0.5-1 J/kg. If an intravenous line is already in place (antecubital preferred over a hand vein), an IV bolus of 0.1-0.2 mg/kg adenosine may be given prior to cardioversion. Adenosine causes a transient AV block and sinus bradycardia, thus interrupting the reentrant circuit involving the AV node and accessory pathway. It must be remembered that this medication is metabolized by the red blood cells and has a very short half-life (approximately 5 seconds), therefore it should be administered via bolus injection followed by an immediate bolus of saline (rapid push and flush). A 12-lead electrocardiogram should be obtained before and after conversion, if possible, and a rhythm strip should be continuously run during attempted conversion. External pacing equipment should be available since some patients go into sinus arrest following administration of adenosine. Potential side effects with adenosine include hypotension, bronchospasm, and flushing.
If adenosine initially fails to convert the SVT, but the patient is hemodynamically stable, they may be started on a medication such as propranolol, digoxin or verapamil (digoxin should be avoided in WPW, verapamil should be avoided in infants) followed by a repeat dose of adenosine. Transesophageal or transvenous pacing is reserved for refractory cases. It is important to remember not to use digoxin on patients with ventricular pre-excitation (e.g., WPW), as it may increase antegrade conduction down the accessory pathway. Patients with WPW are more prone to develop atrial flutter or fibrillation, and are therefore at risk for 1:1 conduction to the ventricles while on digoxin, potentially sending the patient into ventricular tachycardia or fibrillation and can pose a risk of sudden death.
Long-term management of SVT depends on the severity and frequency of episodes. In those patients with no ventricular pre-excitation and infrequent, mild episodes that can be converted with vagal maneuvers, no treatment is required. Patients with frequent or prolonged episodes, or severe symptoms, should be started on a beta-blocker or digoxin, or rarely calcium channel blocker. Patients diagnosed in infancy often will not require continued treatment beyond 1 year of age, but may have recurrent episodes later in life (around 5-8 years of age). With the presence of severe symptoms, refractory SVT, or other situations (e.g., patient preference), an electrophysiology (EP) study and ablation can be performed. This process involves mapping out accessory conduction pathways in the heart with the use of electrodes placed in the atria, coronary sinus, and right ventricle through central venous access. Upon localization of the pathway, a specialized ablation catheter is used to burn or freeze the accessory conduction tissue. Patients with WPW syndrome should also undergo an EP study regardless of the presence of SVT to assess their risk of sudden death.
Electrophysiologic studies with subsequent ablation has become standard of care for older children/adolescents with clinically significant SVT. Overall success rates are 90-98%. There are currently two types of ablation: cryo- and radiofrequency ablation. While radiofrequency ablation offers higher cure rates, it also carries increased risk for inducing heart block.
In summary, the majority of cases of SVT occur under 1 year of age, and the prognosis for recurrence at that time is favorable. However, if SVT occurs or persists beyond this point, recurrence is much more likely. Fortunately, death or significant morbidity is rare with the present state of medical management. Most patients can be expected to live a normal life expectancy with little or no lifestyle alteration due to this condition.
OTHER ARRHYTHMIAS
Sinus Arrhythmia: Changes in sinus rhythm (P-wave preceding each QRS complex, with a normal P-wave axis and PR interval) are most often seen with a sinus arrhythmia. In pediatrics, a sinus arrhythmia is secondary to a variation in vagal tone and cardiac preload during the normal respiratory cycle, a normal phenomenon. This causes an increase in heart rate during inspiration and a decrease in heart rate during exhalation.
Sinus bradycardia is a sinus rhythm with a rate below the normal lower limits for age and activity level, and is most often encountered in well-conditioned athletes. Pathologic states in which sinus bradycardia may occur include increased intracranial, intrathoracic, or intraabdominal pressure, and systemic hypertension.
Sinus tachycardia is a sinus rhythm with a rate greater than the normal upper limits for age and activity level. If not activity-related, the tachycardia usually has a secondary cause such as fever, heart failure, pain, anxiety, hypovolemia, anemia, myocarditis, or thyrotoxicosis.
Atrial Ectopic Tachycardia (AET): This type of tachycardia originates from a single atrial focus other than the sinus node. The onset and termination of the tachycardia is gradual and the rate is usually less than AVNRT/AVRT (ranges from 160-240). The ECG shows inverted, or unusually shaped p-waves. May become clinically significant if the overall heart rate is significantly higher than the resting sinus rate. AET can be associated with previous surgeries with suture lines or scars in the atria.
Chaotic Atrial Tachycardia (CAT) / Multifocal Atrial Tachycardia (MAT): Due to multiple automatic foci in atria. Associated with hypoxia/pulmonary disease, atrial disease, theophylline. Gradual onset, with tachycardia only slightly above baseline rates. The ECG shows at least 2 morphologically distinct and abnormal p-waves, with irregular ventricular response, and variable PR, PP, and RR intervals.
Junctional Ectopic Tachycardia (JET) (Figure 3): Common in the immediate post-operative period following corrective surgery for a congenital heart defect involving the region around the AV node (e.g., a VSD or tetralogy of Fallot repair). The ECG typically demonstrates a narrow complex tachycardia with a regular atrial and ventricular rhythm, and a ventricular rate that is more rapid than the atrial rate. This dysrhythmia originates from a focus of enhanced automaticity in the peri-AV nodal region. The heart rate typically rises and decreases gradually (warms up and cools down). This feature helps differentiate it from a reentrant type of tachyarrhythmia.
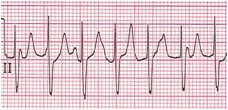
Figure 3. Junctional Ectopic Tachycardia
Atrial flutter (Figure 4) may present with a regular or regularly irregular tachycardia with an atrial rate in the range of 250 to 400 beats per minute. The classic sawtooth flutter waves may be seen, or revealed following a dose of adenosine. The ventricular rate will depend on the degree of A-V conduction (e.g. 2:1, 3:1, etc.). Atrial flutter will most often be encountered in the setting of congenital heart disease, presence of significant mitral/tricuspid valve regurgitation with atrial dilatation, myocarditis, but can also be seen in otherwise healthy newborns.
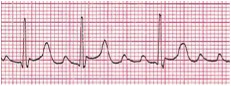
Figure 4. Atrial Flutter
Atrial fibrillation (Figure 5) demonstrates a rapid atrial rate (300-500 beats per minute) with a very chaotic pattern, and an irregularly irregular ventricular rhythm. Atrial fibrillation is most often seen in older children following palliative surgery for congenital heart defects (especially those involving intra-atrial surgery) and in children with significant atrioventricular valve disease.
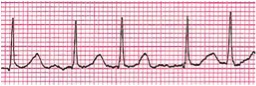
Figure 5. Atrial Fibrillation
Significant ventricular arrhythmias, such as ventricular tachycardia (VT) (Figure 6) and ventricular fibrillation (VF), are rarely encountered in the pediatric age group except in association with certain post-operative conditions. Premature ventricular contractions (PVC) are not uncommon in infants, older children, and adolescents, and they are considered completely benign and do not necessitate any intervention. Patients with ventricular arrhythmias may be asymptomatic or they may present with symptoms of palpitations, chest pain, dizziness, and/or syncope. Ventricular tachycardia is defined at 3 or more consecutive abnormal QRS complexes at a rate greater than 120 beats per minute. As mentioned previously, SVT may occasionally present as a wide complex tachycardia, which may be difficult to distinguish from ventricular tachycardia. In these cases, the definitive diagnosis may not be known until the patient is converted to a sinus rhythm. In these situations the patient should be presumptively treated as having ventricular tachycardia (VT) until proven otherwise. It should be remembered that VT does not always present as a wide QRS complex tachycardia, especially in infants. Ventricular fibrillation displays unidentifiable QRS complexes due to an uncoordinated state of ventricular depolarization, resulting in a state of poor cardiac output. Significant ventricular dysrhythmias in the pediatric age range are most commonly encountered in the setting of congenital heart disease, myocarditis, cardiomyopathies, myocardial trauma, hypoxia, acidosis, and electrolyte abnormalities (most notably hypokalemia and hyperkalemia).
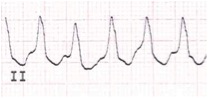
Figure 6. Ventricular Tachycardia
The prolonged QT syndrome causes a distinct type of VT called Torsades de Pointes characterized by a polymorphic VT, which often causes syncope or sudden death. Genetic linkage analyses have isolated a number of genetic foci associated with defects in cardiac ion channels (namely potassium, sodium and calcium channels). Traditionally, patients with long QT syndrome were classified into two groups: Jervell-Lange-Nielsen (autosomal recessive, associated with congenital deafness) and Romano-Ward (autosomal dominant, without deafness); however currently there are more than 15 different long QT syndrome entities associated with different gene mutations. Prolongation of the QT interval may also develop secondary to drugs (anti-arrhythmic agents, antihistamines, antidepressants, antipsychotics, some antibiotics), CNS trauma, cardiomegaly, hypokalemia, and hypocalcemia.
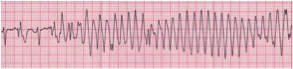
Figure 7. Torsades de Pointes
Various forms of heart block (Figure 8) are usually encountered in children born with heart defects, in heart failure, or may itself be congenital. The congenital form of complete heart block is most commonly seen in the setting of systemic lupus erythematosus or Sjogren's syndrome. In nearly all cases, maternal SS-A/Ro and SS-B/La autoantibodies can be isolated. Conversely, not all fetuses whose mothers are positive for these antibodies will develop heart block. The most common congenital heart defect associated with complete heart block is L-transposition of the great arteries and double inlet left ventricle. If the ventricular rate is too slow to maintain adequate cardiac output, heart failure may develop in utero or postnatally. Treatment involves permanent pacing. The decision to treat depends on the baseline ventricular rate and the likelihood of sudden death. Other potential causes of heart block include medications, myocarditis (viral, Chagas, Lyme, etc.), toxicity (i.e.,(Figure 8) heavy metals).
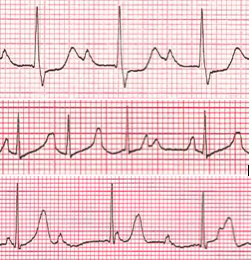
Figure 8. Heart Block
ECG BASICS
Remember that children are not just small adults. HRs, intervals may be faster due to physiology. Also, due to high pulmonary vascular resistance in utero, signs of right ventricular hypertrophy (RVH) are normal in newborns - i.e., rightward axis deviation, dominant R-wave in V1, T-wave inversions (TWI) in V1-V3.
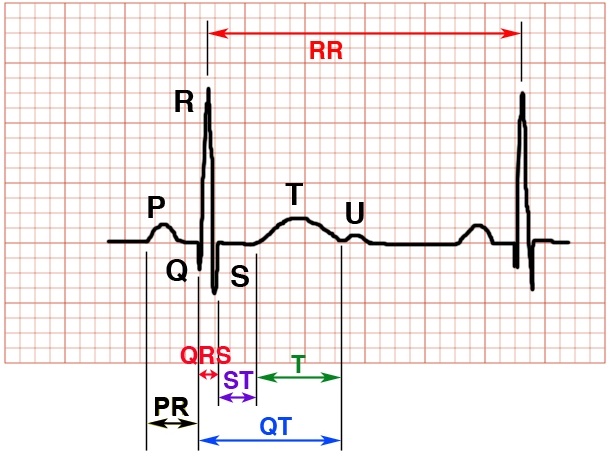
Figure 9. Measuring ECG time intervals
Calibration: Standard 1mV = 10mm and 25mm = 1 sec (at standard speed: 25 mm/sec); 1 big box (5mm) = 0.2 sec, 1 small box (1mm) = 0.04 sec
Rate: Choose two adjacent QRS complexes and count the number of large (5mm) boxes. Heart rate = 300/# of large boxes.
Rhythm: Regular vs. Irregular? Regularly irregular vs irregularly irregular? Is there a p-wave preceding every QRS? Is p-wave positive in leads I, II, aVF and V1?
Axis: Leads I and aVF determine axis. See figure 10 below. Positive deflections in both represent a normal (southeast) axis. Positive Lead I, Negative Lead aVF = left (northeast) axis, seen in AV septal defects (especially endocardial cushion defect), left ventricular hypertrophy (LVH), left bundle branch block (LBBB), and the hypoplastic right heart spectrum including tricuspid atresia. Negative Lead I, Positive Lead aVF = right (southwest) axis. A right axis can be NORMAL in young infants, but is also seen in RVH, tetralogy of Fallot (TOF), atrial septal defect (ASD), R bundle branch block (RBBB). Negative deflections in both leads (northwest axis) may be seen in hypoplastic left heart syndrome (HLHS).
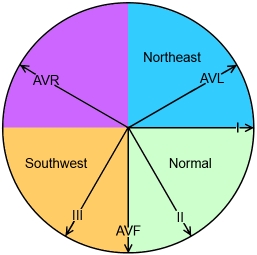
FIgure 10. ECG limb lead and axis quadrants
Intervals
- PR intervals vary based on age and HR: < 8yr old, should be greater than 2, but no more than 4 small blocks. > 8yr old, should be 2.5-4.5 blocks. Longer intervals are seen in heart block. Shorter intervals may be seen in glycogen storage disease and WPW. Variable intervals are seen in Mobitz I (Wenckebach) 2nd degree heart block.
- QRS Should be no longer than 80-100ms (< 2-2.5 blocks). The significance of wide and narrow QRS complexes are discussed above.
- QTc: Bazett method calculated by taking the QT interval (in milliseconds) divided by the square-root of the RR interval (in seconds) of consecutive R waves. Causes of prolonged QTc are mentioned above. Additionally, myocarditis, hypocalcemia and head trauma can cause prolonged QT. Short QTc can be seen in digitalization, hypercalcemia and congenital syndromes. QTc values vary with age: 340-440ms for <10yr olds, <450ms and <460ms for adolescent males and females, respectively.
QRS voltages: Examine V1-V6 for signs of LVH or RVH. Criteria vary based on age (overall, criteria for RVH decrease and criteria for LVH increase as age increases). In general, R-waves in the range of 20-25mm are indicative of ventricular hypertrophy.
P-wave voltages: Normal p-waves should be <3mm in height and <2.5mm in length. In addition to the PR interval and rhythm, as discussed above, right atrial enlargement is present if the P-wave in Lead II is >3mm. Left atrial enlargement is indicated by a wide or notched p-wave.
Questions
1. What is the main difference between accessory pathways in AVNRT and AVRT?
2. What medicine used to treat SVT is contraindicated specifically in WPW?
3. In a hemodynamically stable patient who presents with SVT, what are the two most commonly used methods for attempted conversion to a sinus rhythm?
4. What are two well-known inherited forms of prolonged QT syndrome and their inheritance patterns?
5. In what setting is a "southwest" axis on EKG within normal findings for age?
References
Biondi EA: Focus on Diagnosis: Cardiac Arrhythmias in Children. Pediatrics in Review 2010;31;375-379.
Link MS: Evaluation and Initial Treatment of Supraventricular Tachycardia. N Engl J Med 2012;367:1438-48.
Morillo, CA. Chapter 57 - Supraventricular Tachycardia. In: Crawford MH, DiMarco JP, Paulus WJ: Cardiology, third edition. 2010, Philadelphia: Elsevier, pp 771-784.
Goldberger, Ary. Chapter 22 - How to Interpret an ECG. In: Goldberger's Clinical Electrocardiography: A Simplified Approach, eighth edition. 2013, Philadelphia: Elsevier, pp 214-219.
Salerno JC, Seslar SP: Supraventricular Tachycardia. Arch Pediatr Adolesc Med. 2009;163(3):268-274
Tortoriello TA, Snyder CS, O'Brian Smith E, et al: Frequency of Recurrence Among Infants with Supraventricular Tachycardia and Comparison of Recurrence Rates Among Those With and Without Pre-Excitation and Among Those With and Without Response to Digoxin and/or Propranolol Therapy. Am J Cardiol 2003;92:1045-1049
Answers to questions
1. Accessory pathway in AVNRT is within the AV node, whereas accessory pathway in AVRT is not.
2. Digoxin. In the setting of WPW, digoxin can facilitate impulse conduction via accessory pathway and increase risk for ventricular arrhythmias (i.e., ventricular fibrillation).
3. Vagal maneuvers (ice on the face, or valsalva) and intravenous adenosine.
4. Jervell-Lange-Nielsen (autosomal recessive, associated with deafness) and Romano-Ward (autosomal dominant).
5. Newborns/infants